
-
The Network
-
The Vanguard Study
Latest News and Initiatives
-
 Fred Hutch Leads New Vanguard Study for Cancer Screening Research Network
Fred Hutch Leads New Vanguard Study for Cancer Screening Research NetworkStudy aims to understand the effectiveness and use of multi-cancer detection blood tests.
-
 NCI Selects Two Assays for the Vanguard Study on Multi-Cancer Detection Tests
NCI Selects Two Assays for the Vanguard Study on Multi-Cancer Detection TestsNCI has selected two assays to be included in the Vanguard Study on multi-cancer detection (MCD) tests which will be conducted by the CSRN.
-
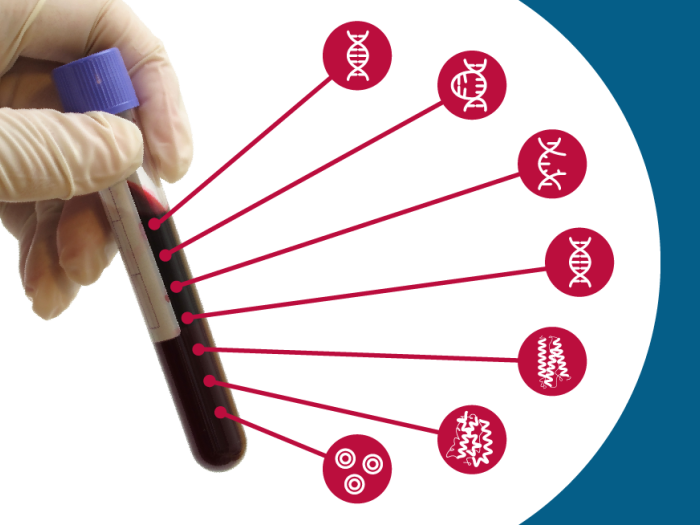 Questions and Answers about Multi-Cancer Detection Tests
Questions and Answers about Multi-Cancer Detection TestsAbout Cancer Screening, About Multi-Cancer Detection Tests, and Risks, Benefits, and Needed Research
Contact CSRN
Email: CSRN@nih.gov
Fred Hutch Cancer Center
The Fred Hutch Cancer Center leads the CSRN multi-center effort to examine the effectiveness of emerging technologies for cancer screening.

Look for this badge at participating Program organizations and institutions. It means they have been awarded a grant by the National Cancer Institute (NCI) as a member of the Cancer Screening Research Network (CSRN).
