Date Posted, by DCP Staff
This issue of Research Highlights focuses on how cells progress to become breast cancer, and separately, how breast cancer may spread. Understanding these transitional moments can help researchers keep invasive cancer from forming, and help us keep invasive cancer from spreading. One study is about mapping the structural and functional patterns inside breast tissue that distinguish between preinvasive and invasive cancer. The second report describes an increased risk of breast cancer spreading in young women diagnosed within 10 years of childbirth.
Study Maps Progression from Precancer to Breast Cancer
In a new study of breast cancer progression, scientists have mapped structural and functional patterns inside breast tissue that may help distinguish between preinvasive and invasive disease.
Using spatial proteomics technology that captures protein positioning within cells, researchers characterized the tumor microenvironment of multiple cell types in the archived breast tissue of women who underwent surgery for ductal carcinoma in situ (DCIS).
Questions about how or if to treat these tiny abnormalities in the milk ducts of the breast, which are increasingly detected by screening mammography, still carries uncertainty. Some DCIS lesions progress to invasive breast cancer, while others do not. But prior research has shown that if left untreated, up to half of DCIS patients will develop breast cancer within 10 years, leading many of them to undergo “presumptive” surgery and other treatments.
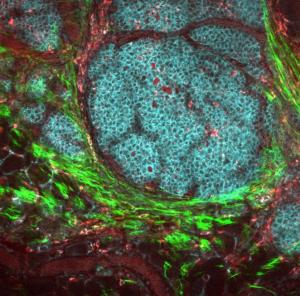
A breast tumor and its microenvironment.
Image from NCI Visuals Online
“Consequently, this [clinical] approach is likely to be overly aggressive for patients [whose cancer does] not progress,” the researchers wrote in Cell. “Thus, understanding what drives DCIS to transition to [invasive breast cancer] is a critical unmet need and opportunity for prevention.”
The study analyzed four transitional states in the tumor microenvironment associated with the shift from DCIS to invasive disease. The research team, which included investigators from Washington University in St. Louis, Stanford University, and Duke University, initially focused on disease progression patterns in 37 proteins of interest. They then compared results in normal breast tissue, tissue from women with DCIS who later developed invasive breast cancer, and tissue from women with DCIS who remained cancer-free.
Altogether, they mapped 16 different cell populations and measured more than 400 features in each sample, all of which came from the Washington University Resource Archival Human Breast Tissue cohort.
The researchers wrote that these findings provide insights into breast cancer progression that could “guide development of future diagnostics and serve as a template for how to conduct similar analyses of pre-invasive cancers.”
This research is from the Human Tumor Atlas Network (HTAN) Consortium, a Cancer MoonshotSM program, and was published in Cell earlier this year as, “Transition to Invasive Breast Cancer is Associated with Progressive Changes in Structure and Composition of Tumor Stroma.”
Study Shows Increased Metastasis Risk in Young Women Diagnosed with Postpartum Breast Cancer
Young women diagnosed with breast cancer within 10 years after childbirth have an increased risk for metastasis, findings from a multicenter cohort study suggest. The statistical analysis, using cases from the Colorado Young Women’s Breast Cancer Cohort, included 701 women aged 45 or younger, who were diagnosed with postpartum breast cancer (PPBC) between January 1, 1981, and December 31, 2014.
Women who had this “underrecognized subset of breast cancer,” the researchers wrote, had a two-fold increased risk for distant metastasis compared with that of women who had not given birth.

This study showed that women diagnosed with breast cancer within 10 years of childbirth were at increased risk of the cancer spreading.
The highest increased risk occurred in women diagnosed with stages 1 and 2 breast cancers, irrespective of hormonal status. In patients with estrogen receptor-positive cancer, increased risk persisted up to 15 years. The findings suggest postpartum status may be an important prognostic indicator for metastasis that should be incorporated into clinical decision-making in the future, according to the researchers.
“In sum, we suggest that poor-prognostic PPBC is an increasing problem that merits more dedicated research,” they wrote. They suggested the incidence of PPBC is likely increasing because older age at first birth correlates with an increased risk for PPBC diagnosis, and more women in the U.S. are opting to delay childbearing.
According to these findings, an estimated 12,000 cases per year in the U.S. would meet the criteria for poor prognostic disease after diagnosis, the researchers wrote. Among the suspected drivers of metastatic risk is cellular remodeling in the breast after childbirth during involution, a physiological event likened to wound healing that increases metastatic efficiency.
Results of the study, supported by grants from the National Cancer Institute, the Department of Defense, and others, were published in JAMA Network Open as, “Association Between Postpartum Breast Cancer Diagnosis and Metastasis and the Clinical Features Underlying Risk.”
If you would like to reproduce some or all of this content, see Reuse of NCI Information for guidance about copyright and permissions. Please credit the National Cancer Institute as the source and link directly to the blog post using the original title, for example: "Research Highlights: Learning More about How Breast Cancer Progresses was originally published by the National Cancer Institute." For questions, contact us at CancerPreventionBlog@mail.nih.gov.
