Date Posted, by DCP Staff
‘Last Mile’ Initiative Will Test Alternative Approach to Screening Through the SHIP Trial Network
The ‘Last Mile’ Initiative SHIP Trial Network was announced today at the White House Cervical Cancer Forum by NCI Director Dr. Kimryn Rathmell.
On January 25, 2024, the National Cancer Institute (NCI), part of the National Institutes of Health, with public and private partners, is launching a new clinical trial network to gather data on a “self-collection” method of human papillomavirus (HPV) testing to prevent cervical cancer. This method, if determined viable, could open new opportunities for improving uptake of cervical cancer screening.
The NCI Cervical Cancer ‘Last Mile’ Initiative is a partnership bringing together multiple federal and private sector stakeholders to contribute evidence on the accuracy and clinical effectiveness of self-collection-based HPV testing for cervical cancer screening.
The initiative is supporting the Self-collection for HPV testing to Improve Cervical Cancer Prevention (SHIP) Trial Network to conduct independent, non-competitive evaluations of the clinical performance of HPV testing using multiple self-collection device and assay combinations that are under review by the Food and Drug Administration (FDA).
Finding Ways to Make HPV Testing More Accessible
Cervical cancer, caused by HPV, is highly preventable by HPV vaccination and by early detection and treatment of cervical precancerous changes. Yet, more than half of all cases of cervical cancer diagnosed in the U.S. occur in individuals who have never been or are infrequently screened.
People are not screened adequately due to reasons as varied as physical and geographic inaccessibility (e.g., residents of rural and urban underserved areas); financial insecurity or lack of health insurance coverage; lack of awareness or understanding of the importance of screening; social, personal, or religious preferences; physical disability and medical conditions; or history of sexual, physical, or psychological trauma that preclude a pelvic examination for sample collection by a clinician.
“The ‘Last Mile’ Initiative has the potential to add self-collection as a screening modality to reach at least some of these underserved and underscreened individuals in the U.S.,” said NCI Principal Deputy Director Douglas R. Lowy, M.D.
The current standard approach to cervical screening involves clinic visits for specimen collection via pelvic examinations performed by clinicians. As an alternative, individuals could collect vaginal samples at home and send them in for HPV testing. Self-collection could also be offered during other health care visits when a pelvic examination is not practical or not preferred.
While the self-collection method has been implemented in other countries, the FDA is currently reviewing the evidence to consider approval of this approach for use in the U.S. Through the ‘Last Mile’ Initiative, NCI has been engaged in discussions with the FDA’s Center for Devices and Radiological Health (CDRH) and industry partners who have current FDA-approved assays for primary HPV testing by a clinician-collected sample.
“The SHIP Trial will generate independent clinical evidence on accuracy, usability, acceptability, and effectiveness to inform the FDA’s reviews for extending current regulatory approvals to include self-collection as an alternative sample collection approach,” said Vikrant Sahasrabuddhe, M.B.B.S., Dr.P.H., of the NCI Division of Cancer Prevention (DCP), who will co-lead the SHIP Trial.
The SHIP Trial design has been integrated with the regulatory pathways recommended by the FDA, and NCI expects SHIP Trial data to be submitted on a rolling basis to inform the FDA’s review and regulatory actions. Industry partners can refer to this data for both pre-approval and post-approval submissions to fulfill regulatory requirements.
A Nationwide Research Network
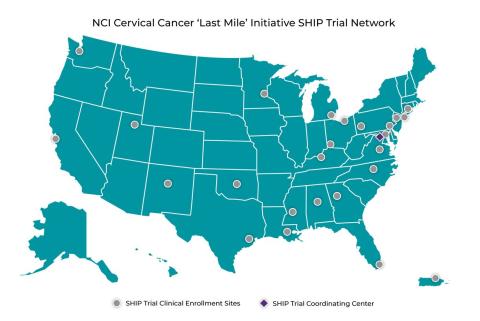
The SHIP Trial Network is comprised of a Coordinating Center at the NCI Frederick National Laboratory for Cancer Research (FNLCR) operated by Leidos Biomedical Research, Inc., in Frederick, Maryland, and a multi-center infrastructure supported through 25 Clinical Enrollment Sites across the U.S. (listed below). The Coordinating Center will facilitate protocol development, onboard Clinical Enrollment Sites, and provide centralized support for ethics review, data management, quality assurance monitoring, specimen shipment and logistics, and specimen repositories. It is expected that participant enrollment will commence by the second quarter of 2024.
“The SHIP Trial Clinical Enrollment Sites cover a wide spectrum of health system settings and have the potential to enroll geographically, socioeconomically, racially, and ethnically diverse participants. Demonstrating that underserved populations are willing to participate in self-collection-based cervical cancer screening could help us reach those populations with the greatest need,” said DCP Director Philip Castle, Ph.D., M.P.H., who will co-lead the SHIP Trial with Dr. Sahasrabuddhe and principal investigators at the Clinical Enrollment Sites.
NCI is also partnering with professional societies to revise and update clinical practice guidelines for integrating self-collection approaches into clinical practice after regulatory approvals. In addition, NCI is developing a publicly accessible repository of self-collection instructions and educational materials for clinicians, community health workers, and people who may use the testing method in the future.
SHIP Trial Coordinating Center
- NCI FNLCR operated by Leidos Biomedical Research, Inc., Frederick, Maryland
SHIP Trial Clinical Enrollment Sites
- Emory University School of Medicine, Atlanta
- Rutgers New Jersey Medical School, Newark, New Jersey
- University of Michigan Medical School, Ann Arbor, Michigan
- University of North Carolina at Chapel Hill School of Medicine, Chapel Hill, North Carolina
- University of Texas MD Anderson Cancer Center, Houston
- University of Oklahoma Stephenson Cancer Center, Oklahoma City
- University of New Mexico Health Sciences Center, Albuquerque, New Mexico
- University of Pittsburgh Medical Center Magee-Womens Hospital, Pittsburgh
- University of Alabama at Birmingham School of Medicine, Birmingham, Alabama
- Johns Hopkins University School of Medicine, Baltimore
- Louisiana State University Health Sciences Center, New Orleans
- University of Miami Sylvester Comprehensive Cancer Center, Miami
- Virginia Commonwealth University School of Medicine, Richmond, Virginia
- Weill Cornell Medicine and New York Presbyterian Hospitals, New York
- Cleveland Clinic Ob/Gyn and Women’s Health Institute, Cleveland
- University of Washington School of Public Health, Seattle
- University of Puerto Rico Comprehensive Cancer Center, San Juan, Puerto Rico
- Minneapolis VA Healthcare System, Minneapolis
- Yale New Haven Health, New Haven, Connecticut
- University of Pennsylvania Abramson Cancer Center, Philadelphia
- University of Mississippi Medical Center, Jackson, Mississippi
- University of Utah Huntsman Cancer Institute, Salt Lake City
- University of Cincinnati College of Medicine, Cincinnati
- University of Louisville School of Medicine, Louisville, Kentucky
- University of California San Francisco School of Medicine, San Francisco
Related links
- NCI Cervical Cancer ‘Last Mile’ Initiative
- Cancer Healthcast: NCI ‘Last Mile’ Initiative Innovates Self-Sampling for HPV Screening
- Relevant publications:
- Primary Human Papillomavirus Testing and Other New Technologies for Cervical Cancer Screening. Einstein MH, Zhou N, Gabor L, Sahasrabuddhe VV. Obstet Gynecol. 2023 Nov 1;142(5):1036-1043. PMID: 37708516
- HPV testing of self-samples: Influence of collection and sample handling procedures on clinical accuracy to detect cervical precancer. Arbyn M, Latsuzbaia A, Castle PE, Sahasrabuddhe VV, Broeck DV. Lancet Reg Health Eur. 2022 Feb 17;14:100332. PMID: 35243475
- Meta-analysis of agreement/concordance statistics in studies comparing self- vs clinician-collected samples for HPV testing in cervical cancer screening. Arbyn M, Castle PE, Schiffman M, Wentzensen N, Heckman-Stoddard B, Sahasrabuddhe VV. Int J Cancer. 2022 Jul 15;151(2):308-312. PMID: 35179777
- Primary Human Papillomavirus Testing and Other New Technologies for Cervical Cancer Screening. Einstein MH, Zhou N, Gabor L, Sahasrabuddhe VV. Obstet Gynecol. 2023 Nov 1;142(5):1036-1043. PMID: 37708516
If you would like to reproduce some or all of this content, see Reuse of NCI Information for guidance about copyright and permissions. Please credit the National Cancer Institute as the source and link directly to the blog post using the original title, for example: "NCI Launches Network to Study Self-Collection for HPV Testing to Prevent Cervical Cancer was originally published by the National Cancer Institute." For questions, contact us at CancerPreventionBlog@mail.nih.gov.
