Date Posted, by Jack J. Lee, Ph.D.
Lung cancer, the leading cause of cancer deaths worldwide killing 1.8 million people each year, is often diagnosed at an advanced stage when the chances for a cure are limited.
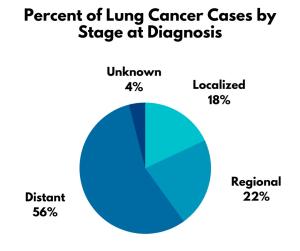
In the United States, almost 60% of people diagnosed with localized lung and bronchus cancer are likely to survive for 5 years. This is nearly 10 times more than those who are not diagnosed until their cancer has spread to distant parts of the body, who have a 5-year relative survival of 6.3%. Right now, more than half of lung cancers are diagnosed at this late state.
Percent of Lung Cancer Cases by Stage at Diagnosis
Researchers are working to change that with a precision approach to early detection.
“Detecting lung early is very important for decreasing mortality,” said Jennifer Beane, Ph.D., a computational biologist at Boston University School of Medicine. “If you detect lung cancers early, you can resect them for cure.”
The U.S. Preventive Services Task Force recommends annual screening by low-dose computed tomography (CT) for adults aged 50 to 80 years with a 20 pack-year smoking history (averaging a pack of cigarettes a day for 20 years) who currently smoke or have quit within the past 15 years. But these criteria don’t cover everyone who develops lung cancer.
“Only 25% to 30% of patients who have lung cancer would have been captured by the current lung cancer screening guidelines,” said Ehab Billatos, M.D., a pulmonologist at Boston University School of Medicine. “We're missing a lot of patients.”
The uptake of low-dose CT for lung cancer screening also has been low, with some estimates that fewer than 5% of those eligible to be screened are getting the tests. Additional tests that can measure molecular markers in blood or tissue may help to improve screening rates.
Drs. Beane and Billatos are searching for new biomarkers to detect lung cancer at earlier stages. Earlier detection could translate into more effective treatment for patients. “If we can just keep pushing the envelope earlier and earlier, then our chance of enhancing people's lives is better,” Dr. Beane said.
The Fields of Injury and Cancerization
Cancer-causing agents such as smoking, or environmental exposures such as radon, damage airway cells, some of which are the specific cells that form tumors. “You could think of it as that there's an injury of some sort to the lung,” Dr. Billatos said. “When you injure something in the lung, then you can measure what the response to that injury is or how bad that injury is.”
Investigating the Field of Injury in the Lung and Airway
These molecular changes can create a “field of injury” that can make a person more susceptible to developing lung cancer. There is also a second field localized to where tumors form. A tumor can affect nearby cells and cause molecular alterations, such as turning up or turning down the expression of certain genes. This concept is known as “field cancerization.”
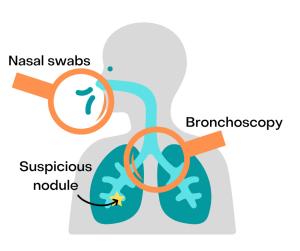
These field effects can inform lung cancer diagnosis. In 2015, researchers identified a molecular signature for lung cancer in current and former smokers in cells obtained by brushing the main bronchi, the passages that branch from the windpipe. They discovered molecular changes reflecting cancer development in these regions, which are more easily accessible than the smaller airways buried deep within lung tissue where lung cancer often develops. As only a small fraction of suspicious pulmonary lesions are lung cancers, the molecular changes can be used to identify pulmonary nodules likely to be benign and reduce the number of invasive diagnostic procedures. The results led to the development of a commercial test, Percepta®, which is used to rule out the presence of lung cancer in patients undergoing bronchoscopy for suspicion of lung cancer.
A 2017 study found that the field effect extends even further into the nasal epithelium. Gene expression profiles of cells obtained by nasal swabs also contain molecular changes associated with lung cancer. Nasal swabs are very easy to obtain and, in the future, could be used as part of lung cancer screening to help figure out which screen-detected nodules are likely to be lung cancer. These studies were supported through the NCI’s Early Detection Research Network (EDRN).
Immune System Changes
Researchers also are trying to identify the early molecular changes in airway cells that are associated with the presence of precancerous changes in the lung. The hope is that by understanding the very earliest changes that precede the development of lung cancer that scientists can figure out ways to stop these lesions from becoming cancerous, i.e., prevent cancer, not just detect it early.
Dr. Beane and her colleagues have been investigating molecular changes associated with bronchial premalignant lesions (PMLs). These PMLs are precursors for one type of non-small cell lung cancer, squamous cell carcinoma. In a 2017 study, the researchers analyzed bronchial brushings from normal looking tissue in the airways of smokers with and without PMLs. They found that the expression of many genes was altered in patients with PMLs showing that there is an airway “field of injury” associated with these very early changes.
In a more recent study, Dr. Beane and colleagues analyzed the bronchial PMLs themselves and found that the lesions clustered into four distinct subtypes based on molecular characteristics. One subtype included PMLs that tended to look more like lung cancer. A group of these proliferative PMLs persisted in the airway or progressed towards lung cancer and these PMLs had lower expression of immune pathways such as interferon signaling and antigen processing/presentation pathways.
“There appears to be an early immune suppression that’s probably at play and causing these lesions to progress towards cancer,” Dr. Beane said. “Our research focus is now aimed at trying to understand the causes of the immune suppression.” The group is part of a large NCI-sponsored consortium called the Human Tumor Atlas Network and is developing a Lung Pre-Cancer Atlas to provide a detailed molecular understanding of precancerous changes in the lung over time as they move toward or away from cancer.
Potentially, these signals in PMLs and the field of injury could serve as biomarkers for people at risk for developing lung cancer. They may also inform future treatments that involve modulation of the immune system at very early stages of disease. “We don’t have anything yet,” Dr. Beane said. “But we’re working on it.”
Finding Lung Cancer Biomarkers for High Risk
Dr. Billatos and his colleagues are evaluating existing airway and blood-based biomarkers for lung cancer in high-risk individuals—and searching for new ones. Several prospective studies are being conducted by the Consortium for Molecular Characterization of Screen-Detected Lesions.
The Detection of Early Lung Cancer Among Military Personnel (DECAMP) consortium was initially led by Dr. Avrum Spira, M.D., M.Sc., a pulmonologist at Boston University School of Medicine, and subsequently Dr. Billatos. The work has received funding from the U.S. Department of Defense and EDRN.
“Leveraging funding from DOD’s grants and resources from EDRN has been a shining example of trans-agency collaboration on lung cancer to detect and assess risk of impending cancer,” said Sudhir Srivastava, Ph.D., M.P.H., chief of the Cancer Biomarkers Research Group in the NCI Division of Cancer Prevention. Dr. Srivastava also served as an advisor to the DOD on lung cancer.
The DECAMP-1 study has recruited 500 smokers with 20 or more pack-years who have indeterminate lung modules detected via imaging scans. These individuals will be followed for up to 2 years until a diagnosis of cancer or benign is made, and the performance of previously established biomarkers will be evaluated. This study has completed recruitment. A new study, DECAMP-1 PLUS, has recently started recruiting and is exploring similar questions in smokers with a smoking history of 10 or more pack-years.
DECAMP-2 is monitoring participants who smoke or used to smoke and are at high risk of developing lung cancer. By following individuals over 4 years, the team is hoping to discover new molecular markers as lung cancers develop. The study is collecting a variety of biosamples including blood, urine, nasal brushings, cheek brushings and bronchial brushings.
“There's a lot of different specimens that we're trying to get,” Dr. Billatos said. The aim is to use specimens that are easy to collect while still being able to identify people with cancer. Dr. Billatos is an EDRN co-investigator and presented details about DECAMP during the 37th Steering Committee Meeting of the EDRN in October 2021.
“It's very hard to acquire this type of data,” Dr. Billatos said. The vision is to provide the data as a resource for the community. “People can investigate for biomarker development. And not just these independent biomarkers—there’s also the potential to integrate multiple tests.”
References
Beane JE, Mazzilli SA, Campbell JD, et al. Molecular subtyping reveals immune alterations associated with progression of bronchial premalignant lesions. Nature Communications 2019; 10(1):1856.
Billatos E, Vick JL, Lenburg ME, Spira AE. The Airway Transcriptome as a Biomarker for Early Lung Cancer Detection. Clinical Cancer Research 2018; 24(13):2984-2992.
Billatos E, Duan F, Moses E, et al. Detection of early lung cancer among military personnel (DECAMP) consortium: study protocols. BMC Pulmonary Medicine 2019; 19(1):59.
If you would like to reproduce some or all of this content, see Reuse of NCI Information for guidance about copyright and permissions. Please credit the National Cancer Institute as the source and link directly to the blog post using the original title, for example: "Identifying New Biomarkers to Detect Lung Cancer Earlier was originally published by the National Cancer Institute." For questions, contact us at CancerPreventionBlog@mail.nih.gov.
