Date Posted, by Susan Jenks
In a new NCI-sponsored study now recruiting participants, investigators will evaluate two different screening strategies for non-cancerous pancreatic cysts to determine which works best at detecting early malignancy.
Even though few of these sac-like structures are likely to develop into pancreatic cancer, researchers said, accurately predicting which cysts pose the highest cancer risk is essential to improving prevention and early detection.

David Weinberg, M.D.
“These surveillance strategies have never been tested prospectively before for their clinical benefit or relative costs and benefits,” said David Weinberg, M.D., chairman of medicine at Fox Chase Cancer Center and the principal investigator of the large clinical trial being conducted by the ECOG-ACRIN Cancer Research Group with support from the NCI. The study EA2185 is comparing the two standard follow-up screening schedules currently in use by physicians. Researchers do not know which of the two is best for patients.
“The problem is we don’t want any patients to slip through our fingers” who might otherwise develop one of these often fatal, difficult-to-treat cancers, Dr. Weinberg said. Pancreatic cancers account for 3% of all cancers in the United States and 7% of all cancer deaths.
Screening for pancreatic cysts has long presented clinicians and patients with the challenging dilemma of whether benefits outweigh the risks. Finding pancreatic cancers early when they are most treatable can improve relative overall 5-year survival. But, studies have shown that the surgical removal of these cysts carries an estimated 2% mortality rate or higher, especially in older individuals, and a 30% chance for complications following surgery.
“If you take all comers with pancreatic cysts, the risk they will turn cancerous is about 0.25% per year,” Dr. Weinberg said. Given the dire prognosis for many patients with these cancers, he said, that’s not trivial, and it illustrates the tension doctors labor under to find these cancers much earlier.
“Even with the best of intentions and the very best imaging we have now,” Dr. Weinberg said, only about 50% of patients will have either cancer or high-grade dysplasia, a precursor to cancer development, when they go to the operating room. So, the remaining 50% endure the worry, cost and risks without apparent benefit, he said.
How Common are Pancreatic Cysts?
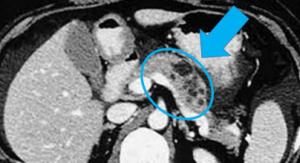
- Cysts can occur at any age, but are more common as we get older.
- Up to 7 million people over age 60 in the U.S. may develop a pancreatic cyst that is large enough to need imaging surveillance.
In this image, the pancreas is circled and shows several cysts of varying sizes.
The screening trial aims to address these concerns. Eventually 4,606 asymptomatic individuals with newly identified pancreatic cysts will be randomized to surveillance strategies based on either the 2012 Fukuoka guidelines (from the 14th meeting of the International Association of Pancreatology in Fukuoka, Japan), or to the 2015 American Gastroenterological Association (AGA) evidence-based guidelines (PDF, 103 KB).
Eligible participants between the ages of 50 and 75 will be followed for at least 5 years as they undergo cross-sectional imaging with magnetic resonance imaging (MRI) or computed tomography (CT), and endoscopic ultrasound (EUS) for signs of early disease.
The main difference between the two screening approaches, Dr. Weinberg said, lies in screening frequency, with the Fukuoka surveillance system having more frequent patient monitoring, while the AGA system calls for less frequent monitoring of patients’ pancreatic cysts.
In the less frequent surveillance arm of the trial, which adheres to the AGA’s guidelines, patients undergo MRI or CT at the beginning of the trial and again in 1 year. In the more frequent Fukuoka surveillance arm, in comparison, patients whose cysts measure 1 to 2 centimeters will undergo MRI or CT every 6 months for 1 year, then every 12 months for 2 years, and then every 24 months thereafter. At later timepoints for each arm, EUS is added and MRI and CT repeated at various intervals.
During the trial, doctors will recommend surgery to any participant in the study who develops presumed risk factors for a cancerous lesion, such as jaundice in the skin and eyes due to a pancreatic obstruction; specific changes in the cyst wall; or a drainage problem in the main pancreatic duct.
Uncovering How Cysts Behave
“By far, we’re operating on more of these cysts than we need to,” acknowledged Kevin Soares, M.D., a cancer surgeon at Memorial Sloan Kettering Cancer Center. In the general population, a majority of cysts will pose no problems for most patients, he said, but a recent three-fold up-tick in detection—due to improved technologies and routine scanning in the nation’s emergency departments—means clinicians need to do better at distinguishing the high-risk ones.
At Memorial Sloan Kettering, patients diagnosed with pancreatic cysts undergo regular monitoring for rapid changes in size or other features, such as high-grade dysplasia, that might signal a developing cancer. Now in its fifth year, the surveillance clinic follows the Fukuoka approach, Dr. Soares said, because the AGA guidelines suggest screening should stop after 5 years “and we don’t think the risk necessarily goes away over time.” Even in patients who have had cyst resection, the “rest of the pancreas is still at risk (for cancer) going forward.”
Among the clinical trials in which the hospital participates, is a randomized study looking at how to stop cyst progression to cancer, Dr. Soares said. Participants are first tested for an anti-inflammatory marker identified in cyst fluid, then receive either an anti-inflammatory drug or a placebo to see if there’s a decrease in cyst growth.
At the same time, researchers elsewhere, including the NCI’s Pancreatic Cancer Detection Consortium, are pursuing studies aimed at unraveling the molecular and chemical underpinnings of pancreatic cysts for additional biomarkers to aid in early cancer detection.
For the new EA2185 screening study, an important secondary goal involves the collection and banking of blood, DNA from buccal (cheek) swabs and cyst fluid for future research. “None of the biomarkers we now have are ready for prime time,” Dr. Weinberg said. But by collecting biospecimens over time, they can be assessed later for differences that show a high likelihood for cancer development, he said.
In addition, investigators are using radiomics, a process by which digital medical information is gathered and stored, then exposed to artificial intelligence to look for patterns that the computer can see and the human eye cannot, according to Dr. Weinberg. “It may show us clues about how cysts behave,” he said, as they transform from commonplace entities seen in 20% of the adult population into pancreatic malignancies.
If you would like to reproduce some or all of this content, see Reuse of NCI Information for guidance about copyright and permissions. Please credit the National Cancer Institute as the source and link directly to the blog post using the original title, for example: "Pancreatic Cysts Are Monitored in New Trial Aimed at Reducing Cancer Risk Through Targeted Screening was originally published by the National Cancer Institute." For questions, contact us at CancerPreventionBlog@mail.nih.gov.
