Date Posted, by Susan Jenks
An over-the-counter drug, readily available to consumers for years, may open a new option for preventing colorectal cancer in high-risk individuals.
It's not the mouse that roared but the quiet mouse behind the scenes that inspired researchers to look at the anti-inflammatory drug, naproxen, said Asad Umar, D.V.M., Ph.D., senior advisor to the director for translational research in the National Cancer Institute’s Division of Cancer Prevention and one of the study authors.
In a 2020 study published in Gut and presented at the virtual annual meeting of the American Association for Cancer Research, naproxen was found to work as well as aspirin in preventing colorectal cancer in individuals at high genetic risk for colon cancer. The study also found that beyond reducing inflammation—a hallmark of non-steroidal anti-inflammatory drugs (NSAIDS)—naproxen recruits immune system cells lining the colon, which are vital to the recognition and elimination of developing malignancies.
“This is probably how our natural immune system clears early changes (in the colon) that can lead to colon cancer,” said Eduardo Vilar-Sanchez, M.D., Ph.D., deputy chair of the department of clinical cancer prevention at MD Anderson Cancer Center, and the principal investigator of the naproxen study. “We showed that both doses of naproxen had an impact on the immune response,” he said. This finding could also enhance the success of future cancer preventive vaccine efforts against the inherited disorder Lynch syndrome. The syndrome is associated with five genes with the potential for mutations that increase a person’s lifetime risk of developing colorectal and other cancers.
In one segment of the study, investigators compared the safety of high-and-low dose naproxen to placebo in 80 patients with the syndrome, who were randomized to one of these drugs daily over a 6-month period. As the trial progressed, they also conducted a side-by-side co-clinical trial in mice with equivalent disease, which allowed them to establish similarities in gene expression between mice and humans; to identify a potential biomarker for measuring naproxen activity; and to demonstrate the cancer preventive superiority of naproxen to aspirin, at least in an animal model.
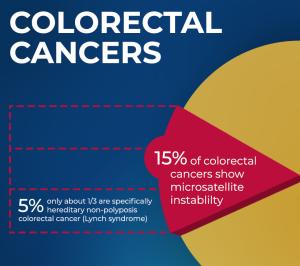
Lynch syndrome is the most common form of hereditary colorectal cancer, affecting some 1.1 million people in the United States. Individuals who inherit it have an estimated lifetime risk as high as 80 percent, compared with 5 percent in the general population, for developing colorectal, endometrial and other cancers at an early age, often before 50 years old. Although frequent colonoscopies, low-dose aspirin, or risk-reducing surgery can decrease cancer incidence in this population, poor compliance with screening surveillance recommendations and the rapid onset of interval cancers between screenings underscores the need for preventive strategies.
Modeling the Molecular Basis of Cancer
Investigators view high risk cancer predispositions such as Lynch syndrome as a powerful human model for understanding the molecular underpinnings of cancers that arise, as this syndrome does, from errors in the DNA mismatch repair genes. Mutations in one of the five known genes for Lynch syndrome (MSH2, MLH1, MSH6, PMS2, and the EPCAM gene) can hinder the body’s natural ability to correct mistakes when copying over genetic information in cells. The mutations cause mistakes in short stretches of DNA repeats known as microsatellites. These errors in the genetic information make the microsatellites unstable and are known as microsatellite instability (MSI). A mutation in any of these genes gives a person an increased lifetime risk of developing colorectal cancer and other related cancers.
Up to 15 percent of all colorectal cancers have MSI due to somatic inactivation of mismatch repair genes, with only a small portion of them, less than 5 percent of all colorectal cancers, occurring in people with Lynch syndrome. But that commonality with other cancers means that successfully intervening in Lynch syndrome may provide insight into cancer prevention in a broader group of individuals whose colorectal cancers develop randomly over time.
“Everything we find in this syndrome is applicable to that larger patient population,” Dr. Vilar Sanchez said. “It could be thousands of patients annually.”
Although naproxen has been readily available to consumers over-the-counter for many years, the decision to test its cancer preventive properties against colorectal cancer now, stems from differing data in mice and in humans about aspirin’s extent of benefits. Many studies have shown a protective effect in people, and the U.S. Preventive Services Task Force suggests a low-dose aspirin regimen may be beneficial, at least in some individuals ages 50 to 69, said Dr. Umar. Mice, on the other hand, have fared poorly. “From other animal models, we already know that there’s a need for huge levels of aspirin in mice to gain any protective effect” against colorectal cancer, Dr. Umar said. That discrepancy with humans made “the mouse behind the scenes the reason we moved to naproxen.”
Unlike aspirin, naproxen remains longer in the body, so lower doses of the drug are needed to achieve a cancer preventive effect. “And, if you take any drug less frequently, it also means the side effects will be less,” Dr. Umar said. “That’s especially important to a cancer prevention trial where we want to minimize toxicities.”
Despite similarities in the study between high- and low-dose naproxen, investigators saw some differences between the two that were dose-dependent. Both doses lowered prostaglandin levels associated with inflammation, as expected, Dr. Umar said, but the higher dose of 400 milligrams brought an increase in gene expression, as well, including in genes involved in cellular immune responses. That finding supports the likelihood investigators will use the higher naproxen dose as an adjuvant in vaccine trials for Lynch syndrome, going forward, now that this NSAID’s safety has been shown. The combination strategy, investigators reason, is a logical next step and holds the most promise for unleashing the power of the immune system against early neoplasms before they turn into cancer.
Accurate diagnosis of Lynch syndrome coupled with the use of risk-reduction strategies, such as surveillance, prophylactic surgery, and cancer prevention, could improve clinical outcomes for colorectal cancer survivors, Dr. Umar said. Yet, despite well-known cancer screening and prevention strategies, there is only a small percentage of Lynch syndrome carriers who know they have this predisposition. The NCI Cancer MoonshotSM is funding research to identify more Lynch syndrome carriers through improvements in genetic testing. This includes recommended universal screening of tumor tissue using MSI testing or the mismatch repair protein immunochemistry in all colorectal or endometrial cancers as both a diagnostic tool for Lynch syndrome, and as a predictive, prognostic, and therapeutic marker for other MSI cancers.
Future research directions will be further identified in upcoming focused meetings of experts in the role of inflammation in carcinogenesis and the role of anti-inflammatories in cancer prevention.
If you would like to reproduce some or all of this content, see Reuse of NCI Information for guidance about copyright and permissions. Please credit the National Cancer Institute as the source and link directly to the blog post using the original title, for example: "Study With Common Pain Drug Shows Change in Immune Biomarkers, Highlights Pathway for Prevention of Colorectal Cancer in People with Lynch Syndrome was originally published by the National Cancer Institute." For questions, contact us at CancerPreventionBlog@mail.nih.gov.