Project Title/Research Areas: Hybrid Plasma Markers that Complement CT Imaging for Early Lung Cancer Detection
Principal Investigator/Institution: Paul Lampe, Ph.D. / McGarry Houghton, M.D., Fred Hutchinson Cancer Research Center
Description of Current Activity
Lung cancer is the leading cause of cancer deaths worldwide with >159,000 deaths annually in the US alone. Making matters worse, 5-year survival rates remain a dismal ~18%, since most lung cancer cases are identified at an advanced stage, which confers a poor prognosis. In contrast, 5-year survival rates are much higher (~56%) when lung cancer is diagnosed at its earliest stages. The National Lung Screening Trial (NLST) employed low-dose Computed Tomography (CT) imaging of the chest to screen for lung cancer in a high-risk population (smokers aged 55-74). This study demonstrated a 20% reduction in mortality in the group receiving CTs when compared to standard care. Unfortunately, pulmonary nodules are a relatively common finding in the lungs of smokers, such that 26% of the subjects required additional work-up for their pulmonary nodules, with <5% of them representing cancer. Analysis of several screening cohorts indicates that 25-50% of smokers >50 years of age have CT identifiable pulmonary nodules but very few of them (~2.5%) are caused by lung cancer. Implementation of recommended screening in VA populations found 56% with nodules but only 1.5% with cancer. Current guidelines for incidentally detected pulmonary nodule evaluation call for additional imaging and/or invasive biopsy procedures depending upon the size/characteristics of the nodule and key clinical parameters (e.g., age, smoking history). There are clinical and economic needs for innovative risk stratification strategies as we begin this new era of recommended screening.
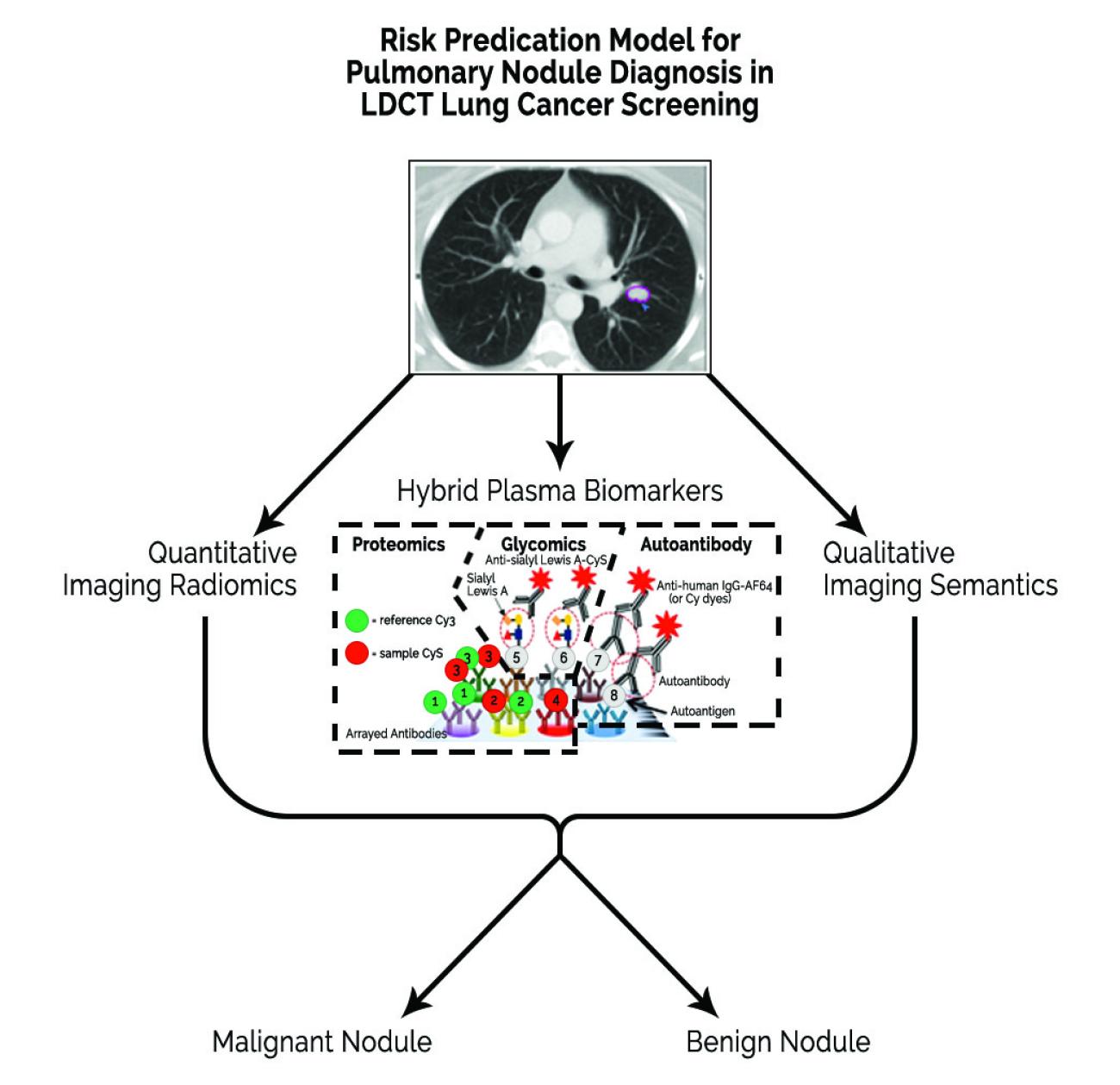
Our project involves the combination of plasma biomarkers with CT image data to improve lung cancer screening. We created a high-density antibody array containing 3200 different antibodies that we use to interrogate pre-diagnostic sample sets from observational trials in a nested case-control design study to evaluate proteomic, glycomic and autoantibody differences. Using pre-diagnostic lung cancer samples from the Cardiovascular Health Study (CHS), we found 30 proteomic, glycomic or autoantibody biomarkers that were significantly increased (p<0.002) in people that are subsequently diagnosed with lung cancer.
Update
As part of this grant, we have used plasma samples from 297 lung nodule positive subjects that have been screened via CT and have known cancer/nodule status (147 were cancer) to test these 30 markers and potentially discover additional candidates (Aim 1, finished). We are currently combining these data with CT imaging parameters and clinical data to create a risk prediction model that we will test in a similar sized prospectively collected cohort (Aim 3). We have also purified B-cells from lung tumor tissue and examined whether autoantibodies present in plasma are tumor-derived to assess their utility for the detection of cancerous nodules (Aim 2, results submitted for publication). We have also begun to add “radiomic” approaches to supplement biomarker and conventional CT image features to improve lung cancer early detection (see figure below).