Project Title/Research Areas: Integrative molecular and imaging approaches for risk of subtype specific breast Subtypes.
Principal Investigator/Institution: Isabelle Bedrosian, M.D. and Randa El-Zein M.D., Ph.D.
Description of Current Activity
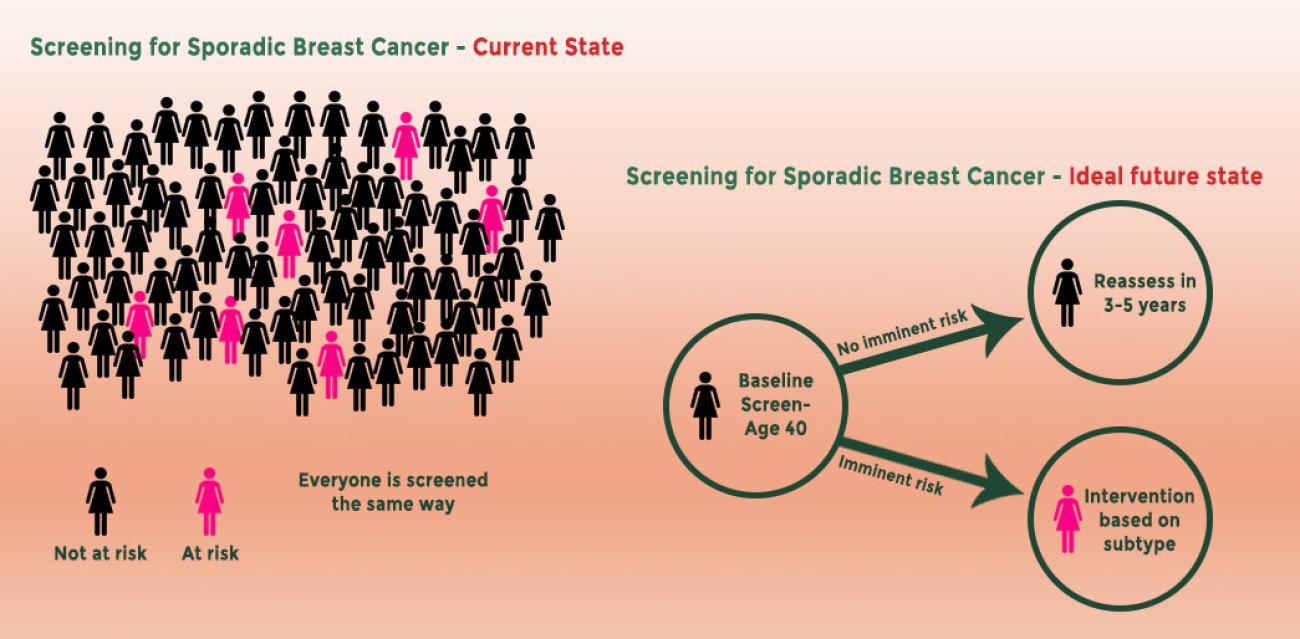
Screen-based early detection remains largely agnostic with regard to risk of breast cancer. In order to maximize the benefit-to-harm ratio of screening programs, it is important to first identify individuals at sufficiently high risk of the disease. The Gail model remains the most widely used tool for the assessment of risk of invasive breast cancer. This model is based on recognized risk factors such as age, family history, personal reproductive history, age at menarche and at first live birth, and medical history, such as number of previous breast biopsies. Although shown to be well-calibrated at the population-level, the Gail model performs poorly at the individual-level. Additionally, this tool has not been validated for patients with atypia nor for minority women, and in these groups, the Gail risk may substantially underestimate true risk. Therefore, for most women, the Gail risk assessment tool is not sufficiently personalized and thus does not provide the most precise risk estimates possible.
Thus for the majority of women, we have come to rely on age as a surrogate of breast cancer risk. This approach has recently led to considerable controversy and it’s clear that many women are being screened unnecessarily. However, the contribution of screen-based early detection in reducing breast cancer mortality is also documented. Therefore, an approach that preserves the power of early detection through screening mammogram but limits overtreatment of women who are NOT at increased risk is needed. Lastly, the heterogeneity of breast cancer has not yet been integrated into early detection and prevention measures. An extensive body of literature over the last decade would suggest that each of the main subtypes of breast cancer have unique biology, unique behavior, and require unique treatment approaches to optimize response. Further, it appears that some of the clinical and demographic risk factors differ between the cancer subtypes. These observations therefore suggest that strategies about risk prediction are likely to be more powerful if they could be tailored to subtype specific risk.
Our study addresses these barriers by testing an approach that integrates computer assisted imaging and blood marker signatures as a subtype; specific early detection tools that could overcome the overutilization of mammography; and expand on the information gained through this screening modality and in turn personalize early detection strategies for women. Since the issue of mammographic screening touches all women in the U.S. at some point in their lives, the impact of personalizing screening guidelines would be substantial.
Aim 1: Create a subtype specific early detection tool by integrating mammographic “premalignant signatures” and blood based biomarkers. We hypothesize that the background breast parenchyma will differ across the subtypes, but in and of themselves these signatures will not be specific enough and we therefore plan to further refine the imaging signatures with blood biomarkers of DNA damage.
Aim 2: To identify the extent of field cancerization associated with the different breast cancer subtypes. We will test the hypothesis that in most patients, breast cancer arises from a background of molecular field change, thus supporting the rationale for developing imaged based premalignant signatures of the breast parenchyma. We will use PAM50 assays to compare the molecular portraits of tissues obtained from four distinct sites within the mammary gland of breast cancer patients.
Aim 3: Determine whether the imaging “premalignant signatures” precede the development of breast cancer. In a subset of women where we have multiple archived antecedent mammograms (n=200), we will test whether the imaging features extracted in aim #1 can be traced back to the mammograms in years prior to development of cancer. We hypothesize that this parenchymal signature derived in aim #1 is detectable in the imaging obtained in the years antecedent to the cancer diagnosis.
Data generated from our study would allow the identification of women that are at imminent risk of not only developing breast cancer, but subtype specific breast cancer, and thus tailor the subsequent intervention.